|
This explains why we can get energy from the starch in potatoes and other plants but not from cellulose, even though both starch and cellulose are polysaccharides composed of glucose molecules linked together. The name derives from its similarity to the oxygen heterocycle furan, but the furanose ring does not have double bonds. They are alpha-D-glucose pyranose beta-D- glucose pyranose and alpha-D-glucose furanose Beta-D-glucose furanose. A furanose is a collective term for carbohydrates that have a chemical structure that includes a five-membered ring system consisting of four carbon atoms and one oxygen atom. The difference between the α and the β forms of sugars may seem trivial, but such structural differences are often crucial in biochemical reactions. 6 membered ring closure generates a stereo genic carbon (chiral carbon) which is called as anomeric carbon that leads to the formation of two diastereomers, preferably known as anomers of glucose. If the hydroxyl group is below carbon number 1 in the sugar, it is in the alpha ( ) position, and if it is above the plane, it is in the beta ( ) position. Any group written to the right in a Fischer projection appears below the plane of the ring in a Haworth projection, and any group written to the left in a Fischer projection appears above the plane in a Haworth projection. Glucose in a ring form can have two different hydroxyl group arrangements (OH) around the anomeric carbon (carbon 1 that becomes asymmetric in the ring formation process). 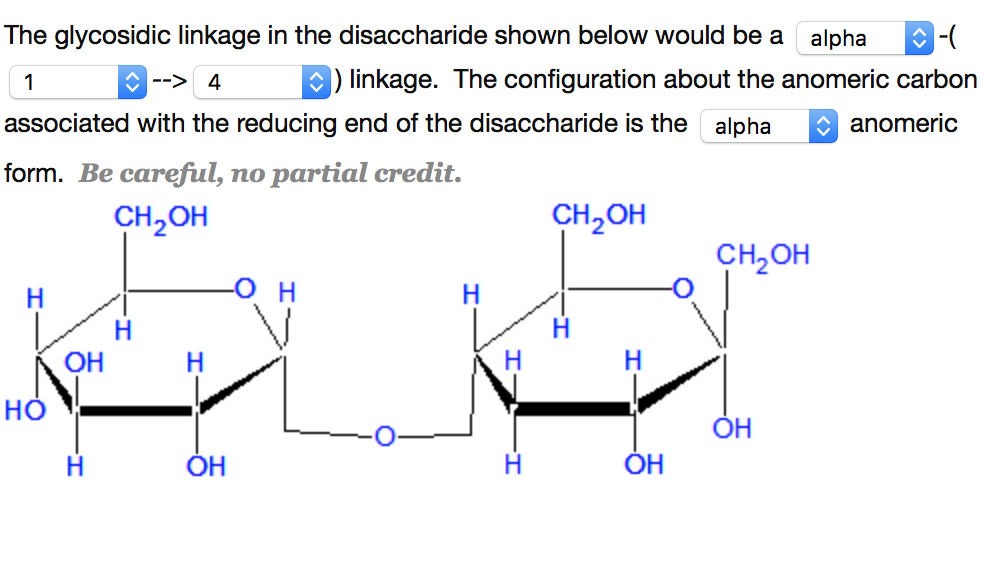
The structure is simplified to show only the functional groups attached to the carbon atoms. The molecules are drawn as planar hexagons with a darkened edge representing the side facing toward the viewer. A sugar is in its -anomer form when the anomeric carbon’s substituent is on the opposite side of the plane as the highest numbered chiral center’s substituent. Be aware that a cyclic sugar can exist in two possible anomers: an -anomer and a -anomer. 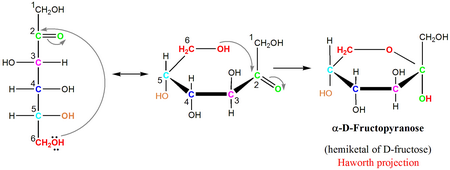
The a anomer is formed when the hydroxyl group bonded to the anomeric carbon is trans (opposite side) to the carbon outside the ring (carbon 6). In a cyclic sugar, the anomeric carbon is the carbon that has two bonds to oxygen. Both carbons differ in configuration of the hydroxyl group. As shown in the structure above a and b are present at adjacent to oxygen atom. The two possible arrangements, termed anomers, are referred to as the alpha () and beta () anomers. Carbon adjacent of oxygen atom in the cyclic structure of glucose or fructose is known as anomeric carbon. The sucrose molecule is the only one of the common disaccharides that have an alpha-1,beta-2-glycosidic established between carbon 1 in glucose and carbon 2 in fructose. \)) the cyclic forms of sugars are depicted using a convention first suggested by Walter N. The anomeric carbon is the carbonyl carbon that reacts to form the cyclic structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
